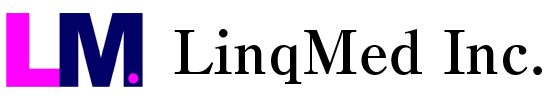Innovative “Visible” Cancer Treatment: Our Challenge
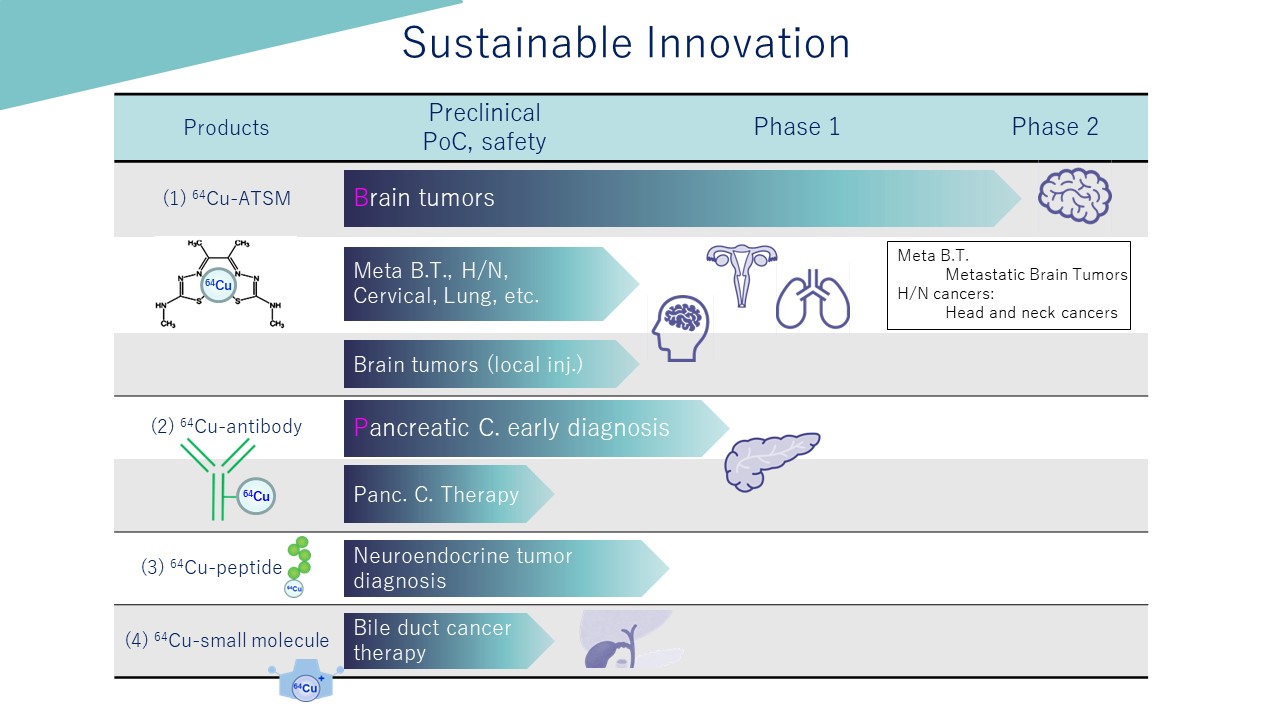
Conventional radiotherapy and chemotherapy for cancer have had problems such as insufficient therapeutic effects and significant side effects on healthy cells. On the other hand, we have developed a radiopharmaceutical employing copper radioisotope, 64Cu, which can overcome these problems. In addition to the beta particles used in conventional radiotherapeutics, 64Cu emits a unique radioactive ray called Auger electron, which enables us to treat cancer cells effectively with high energy. Moreover, as 64Cu is a positron-emitting isotope, it allows us to proceed with treatments while we visibly and noninvasively trace the drug accumulation using Positron Emission Tomography, PET scan.
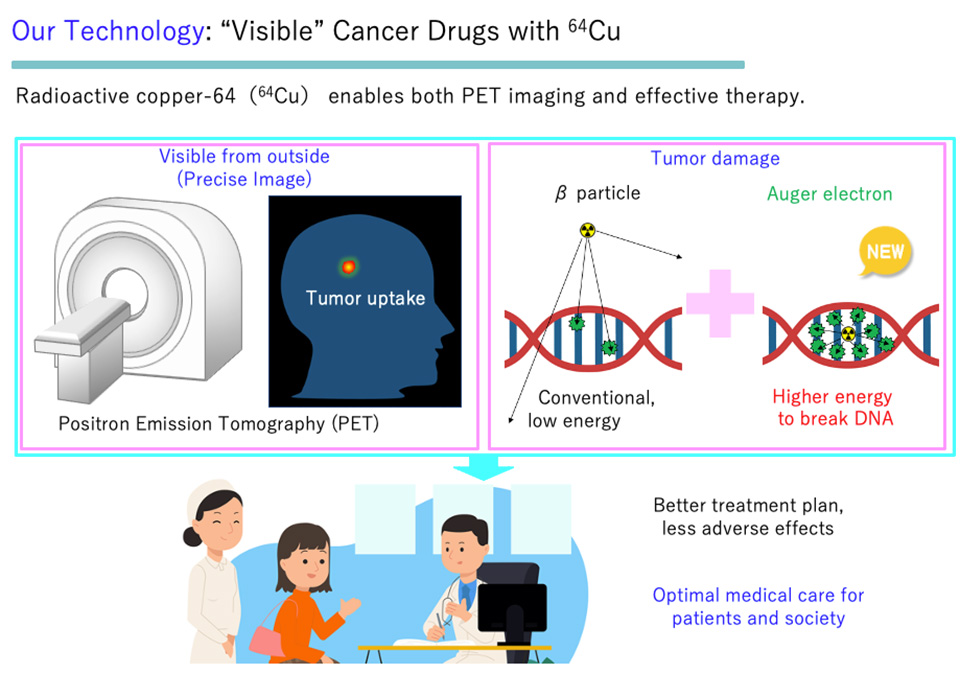
Furthermore, 64Cu can be bound to various molecules that exhibit high affinity with cancer, making it possible to sustainably create a wide variety of drugs that exhibit cancer-specific effects. By using with 64Cu’s such exceptionalities, we aim to socially implement “Innovative ‘Visible’ Cancer Treatment.”
64Cu-ATSM: The First-in-the-World Clinical Trial of the Radiotherapeutic Drug developed in Japan Underway
As malignant brain tumors cannot be treated adequately with conventional cancer treatment, the development of novel cancer treatment is strongly desired. One known factor for the poor outcome of currently available treatment options is the hypoxic environments of tumor cells, which makes cancer resistant to treatment.
In response to this, we have researched and developed 64Cu-ATSM, which accumulates in tumor cells in a hypoxic environment and exerts a high therapeutic effect. Preclinical studies using cancer cell-xenografted models have shown that 64Cu-ATSM suppresses the growth of malignant brain tumors under hypoxic conditions and improves the survival rate significantly.
Based on this background, 64Cu-ATSM is expected to become a new therapeutic agent for malignant brain tumors, and a phase 1 clinical trial of 64Cu-ATSM treatment is currently underway. This trial is the first-in-the-world study for the radiotherapeutic drug developed in Japan, and early implementation of this drug to the clinical practice is especially expected.